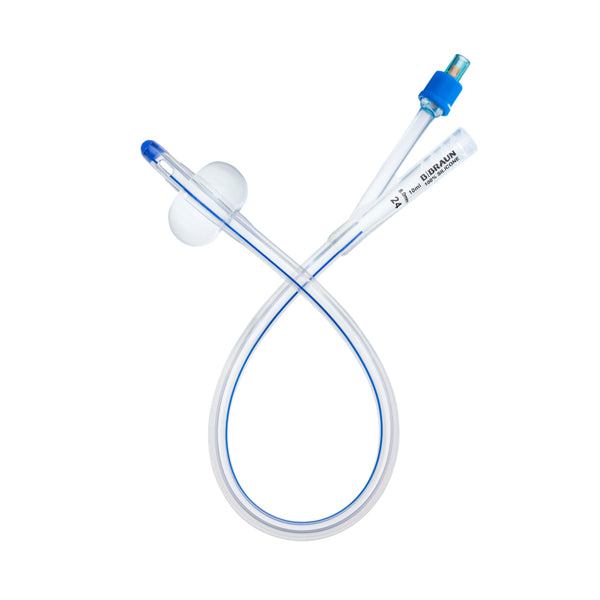
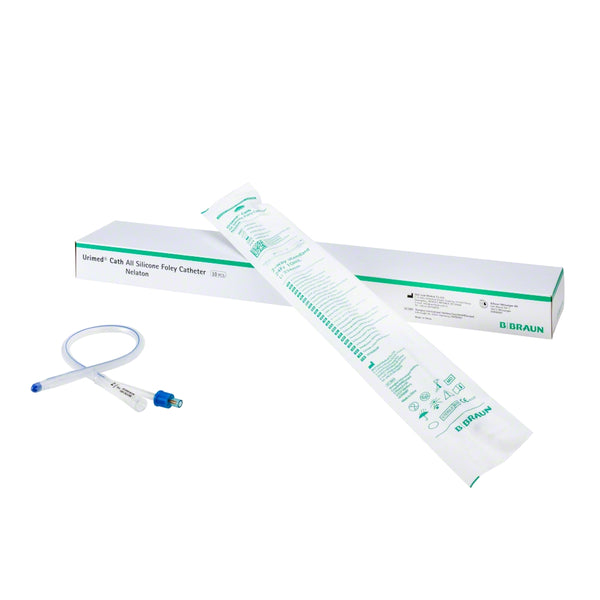
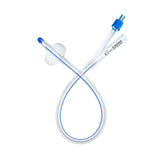
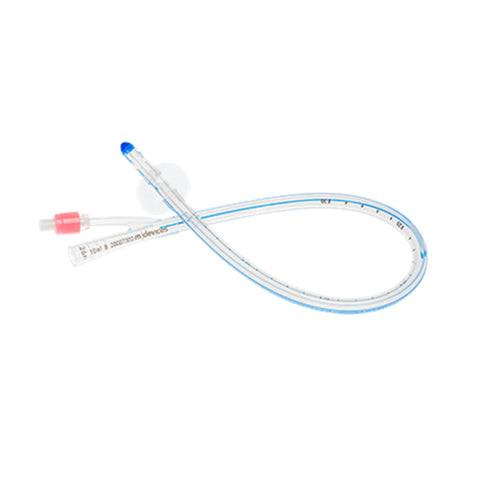
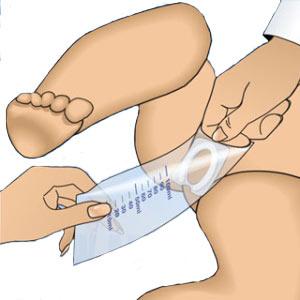
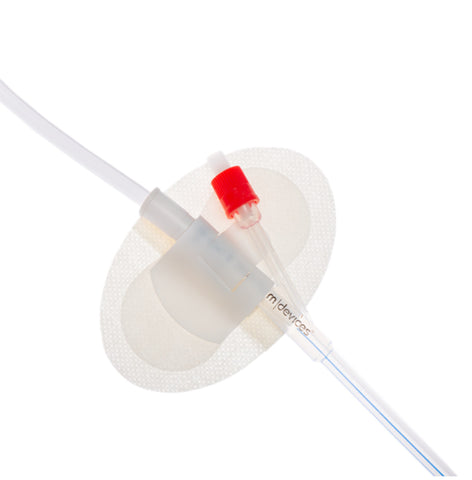
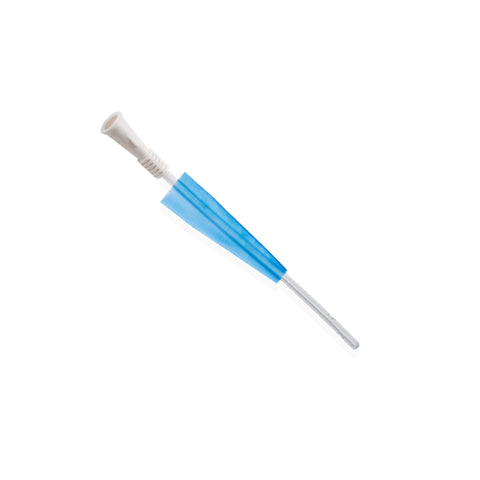
Introducing the Urimed® foley catheter, a two-way silicone foley catheter designed for routine bladder drainage, offering a range of features to enhance patient comfort and clinical efficiency.
Key Features:
- 100% silicone construction, providing a gentle and hypoallergenic solution that minimises tissue irritation and potential damage
- The inner lumen of silicone foley catheters ensures a wider drainage channel compared to latex catheters, enhancing efficiency and reducing trauma during drainage
- Two-way catheter with a length of 42 cm and a 10 ml balloon for effective bladder management
- Latex-free and PVC-free, making it a safe and suitable option for a diverse range of patients
- Radiopaque closed Nelaton tip with 2 drainage eyes for precise placement and effective drainage
- Easy-to-open double sterile packaging ensures aseptic conditions during catheterisation
- Available in a range of sizes from CH12 to CH18, providing flexibility to meet various patient needs
Indications:
- Acute and chronic urinary retention
- Continuous urine outflow for patients with voiding difficulties due to neurological disorders causing paralysis or loss of sensation affecting urination
- Accurate measurement of urinary output in critically ill patients
- Perioperative use for selected surgical procedures
- Urological surgery or other surgery on contiguous structures of the genitourinary tract
- Anticipated prolonged duration of surgery
- Intra-operative monitoring of urinary output
- Healing of open sacral or perineal wounds in incontinent patients
- Long-term immobilisation needs (e.g., potentially unstable thoracic or lumbar spine, multiple traumatic injuries such as pelvic fractures)
- Bladder irrigation
- Continence and skin integrity maintenance (when conservative treatment methods have been unsuccessful)
- Comfort improvement for end-of-life care if needed
- Management of intractable incontinence.
Returns Policy
1. Prior Approval Required: All returns must be approved by us prior to sending any goods back. Please contact us to obtain a return authorisation before returning any items.
2. Claims for Issues: Claims for short delivery, incorrect supply, or damaged goods during transport must be made within 7 days of the delivery date. Please inspect your goods upon receipt and notify us immediately if there are any issues.
3. Return Conditions: We generally do not accept returns for any reason other than a faulty product. This includes returns due to change of mind or incorrect orders However, at our discretion and depending on the product, we may accept non-faulty returns. These will incur a 10% restocking fee based on the product's purchase price. Refunds will be processed for the remainder of the order minus the original shipping cost and restocking fee, after the item has been inspected and approved. The buyer is responsible for returning the item in the same condition it was received. We do not accept returns for items that are damaged, opened, or not in original condition.
4. Lost Returns: If a returned item is lost in transit from the buyer to Solmed, we will not be held responsible. Please ensure that you use a trackable and insured shipping method..
5. TGA Duties: For products regulated by the Therapeutic Goods Administration (TGA), returns must comply with relevant TGA regulations and guidelines. Any return of TGA-regulated products will be subject to additional conditions as required by law.
6. Return Shipping Costs: Return shipping costs are the responsibility of the buyer unless otherwise agreed upon in the case of faulty products.
7. If you have any questions regarding returns or need assistance, please contact us before proceeding with the return process.
Shipping
1. Responsibility for Delivery: Once your parcel is with the courier service, it is beyond our control. We are not responsible for any loss, damage, or delivery issues that may occur once the parcel is in the possession of the courier.
2. Authority to Leave: All orders are dispatched without the requirement of a signature. The courier may leave your parcel in a location they deem to be a “safe place.” We cannot be held liable for any loss, damage, or theft that may arise from this decision.
3.Signature on Delivery: For added security, you may select the Signature on Delivery option at checkout. This ensures your parcel will not be left unattended. If no one is available to receive it, the parcel will be taken to the nearest post office or courier depot for collection.
4. Tracking Information: Tracking numbers will be provided to you via email. It is the buyer’s responsibility to monitor the progress of their parcel using the tracking information provided.
5. Buyer’s Responsibility: Any discrepancies or issues with the delivery must be followed up with the respective courier service by the buyer.
6. Transit Cover (Optional): You may purchase transit cover to insure the value of your order in case of loss, damage, or theft. The cost is 1% of your order total. Please contact us if you’d like to add this to your order. This is the only option that provides coverage for your order value.