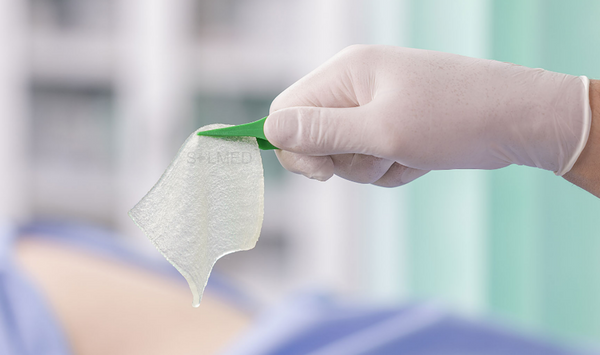
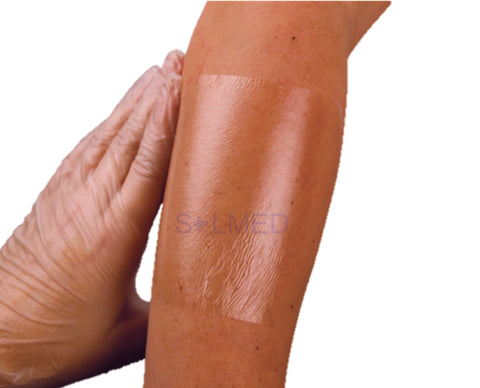
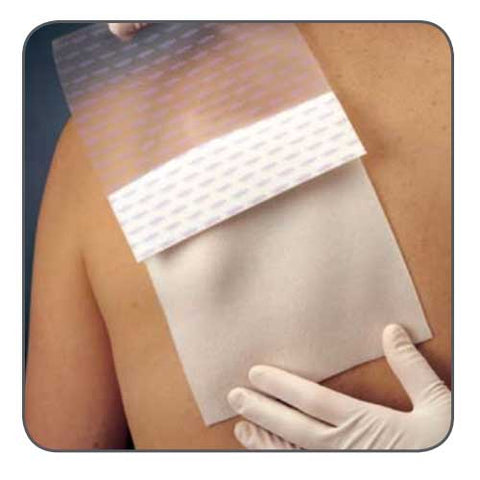
Exufiber is a sterile non-woven dressing made from highly absorbent polyvinyl alcohol fibres. In contact with wound exudate, Exufiber transforms into a gel that facilitates moist wound healing and ease of removal during dressing changes. Exufiber absorbs and retains wound exudate.
Benefits of Exufiber
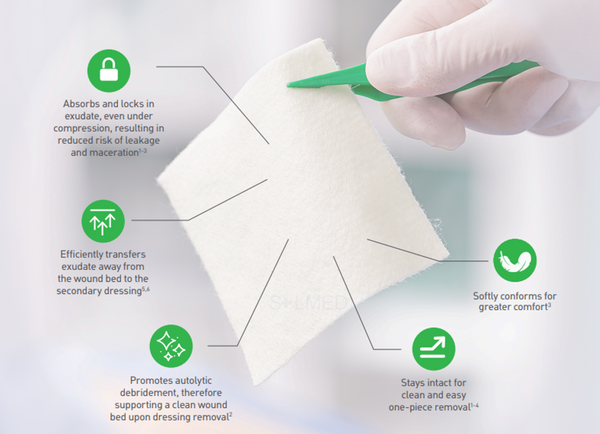
- High tensile strength to enable dressing removal in one piece
- High retention capacity to prevent leakage and maceration
- Absorbs and retains exudate, blood and bacteria
- Highly absorbent, also under compression therapy
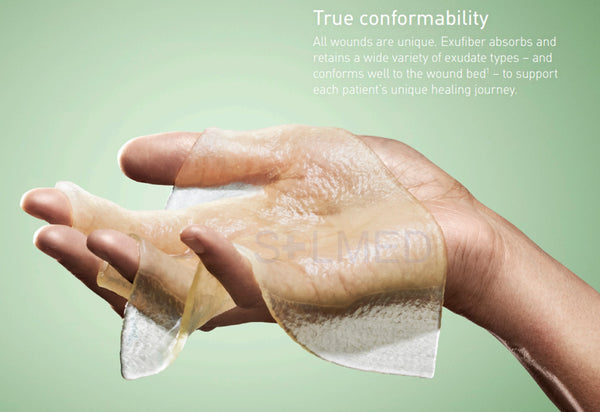
- Soft and conformable which makes it easy to apply
- Hydrolock Technology
- Can be left in place for up to seven days and up to 14 days on donor sites
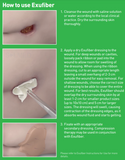
How does it work?
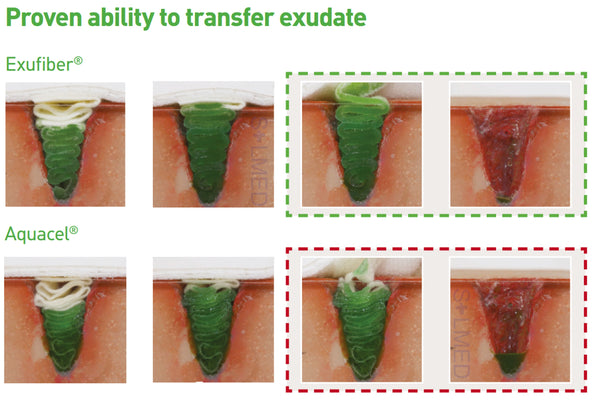
The soft and conformable non-woven polyvinyl alcohol fibres transform into a gel upon contact with exudate. With hydrolock technology the tightly packed fibres absorb and swell when in contact with fluid, keeping exudate locked in, while the capillary action enables transfer of exudate to the secondary dressing. Exufiber promotes autolytic debridement and is removed in one piece, therefore supporting a clean wound bed.
Areas of Use
Exufiber wound dressings are intended to be used on a wide range of exuding and cavity wounds, such as:
- Leg and foot ulcers
- Pressure injuries
- Partial-thickness burns
- Surgical wounds
- Donor sites
- Malignant wounds
Precautions
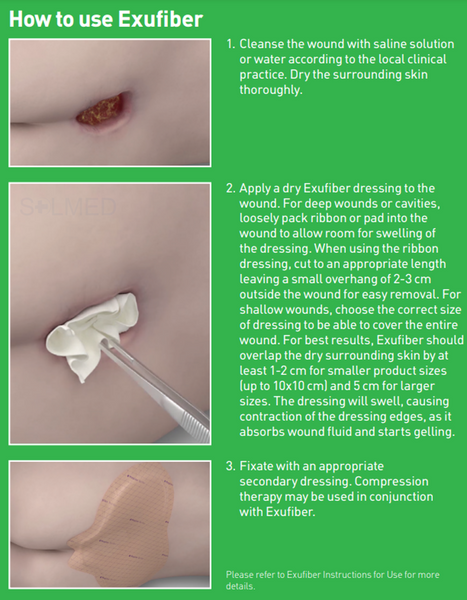
Exufiber is not intended for dry wounds or full-thickness burns. If the dressing dries out and is difficult to remove, it should be moistened according to the local clinical practice and allowed to soak until it lifts easily.
Resources
Returns Policy
1. Prior Approval Required: All returns must be approved by us prior to sending any goods back. Please contact us to obtain a return authorisation before returning any items.
2. Claims for Issues: Claims for short delivery, incorrect supply, or damaged goods during transport must be made within 7 days of the delivery date. Please inspect your goods upon receipt and notify us immediately if there are any issues.
3. Return Conditions: We generally do not accept returns for any reason other than a faulty product. This includes returns due to change of mind or incorrect orders However, at our discretion and depending on the product, we may accept non-faulty returns. These will incur a 10% restocking fee based on the product's purchase price. Refunds will be processed for the remainder of the order minus the original shipping cost and restocking fee, after the item has been inspected and approved. The buyer is responsible for returning the item in the same condition it was received. We do not accept returns for items that are damaged, opened, or not in original condition.
4. Lost Returns: If a returned item is lost in transit from the buyer to Solmed, we will not be held responsible. Please ensure that you use a trackable and insured shipping method..
5. TGA Duties: For products regulated by the Therapeutic Goods Administration (TGA), returns must comply with relevant TGA regulations and guidelines. Any return of TGA-regulated products will be subject to additional conditions as required by law.
6. Return Shipping Costs: Return shipping costs are the responsibility of the buyer unless otherwise agreed upon in the case of faulty products.
7. If you have any questions regarding returns or need assistance, please contact us before proceeding with the return process.
Cancellation Policy
1. Cancellation Request Timeframe: Customers may request to cancel an order within one (1) hour of placing the order. Cancellation requests received within this timeframe will not incur any administration or cancellation fees, provided the order has not yet been processed for fulfilment.
2. Cancellation Requests After One Hour: Cancellation requests made after one (1) hour from the time the order is placed are not guaranteed and will be considered at the sole discretion of Solmed, as products may have already been allocated, processed, or ordered from suppliers for fulfilment.
3. Administration Fee: If Solmed approves a cancellation request submitted after the one (1) hour processing period, a 10% administration fee may be applied to the order value to cover processing, handling, and supplier administration costs.
4. Orders Already Shipped or Ready for Collection: Orders that have already been shipped, dispatched, or marked as ready for collection cannot be cancelled. In these circumstances, the order will instead be subject to our Returns Policy, where applicable.
5. Right to Decline Cancellation: Solmed reserves the right to decline cancellation requests where the order has already entered the fulfilment process, been packed, dispatched, or where the products have been specially ordered or allocated for the customer.
6. How to Request a Cancellation: All cancellation requests must be submitted as soon as possible by contacting us and providing the order number and relevant purchase details. A cancellation request is not considered accepted until confirmed in writing by Solmed.
Shipping
1. Responsibility for Delivery: Once your parcel is with the courier service, it is beyond our control. We are not responsible for any loss, damage, or delivery issues that may occur once the parcel is in the possession of the courier.
2. Authority to Leave: All orders are dispatched without the requirement of a signature. The courier may leave your parcel in a location they deem to be a “safe place.” We cannot be held liable for any loss, damage, or theft that may arise from this decision.
3.Signature on Delivery: For added security, you may select the Signature on Delivery option at checkout. This ensures your parcel will not be left unattended. If no one is available to receive it, the parcel will be taken to the nearest post office or courier depot for collection.
4. Tracking Information: Tracking numbers will be provided to you via email. It is the buyer’s responsibility to monitor the progress of their parcel using the tracking information provided.
5. Buyer’s Responsibility: Any discrepancies or issues with the delivery must be followed up with the respective courier service by the buyer.
6. Transit Cover (Optional): You may purchase transit cover to insure the value of your order in case of loss, damage, or theft. The cost is 1% of your order total. Please contact us if you’d like to add this to your order. This is the only option that provides coverage for your order value.